Properties of water
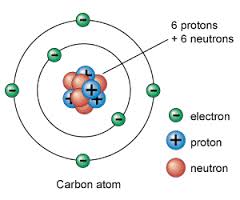
Protons have a positive charge
Electrons have a negative charge
Neutons are neutrally charge (no charge)
A atom consist of protons, electrons, and neutrons. Atoms are usually neutrally charge because the number of protons and electrons are even and a atom is the smallest unit of matter that retains it's properties.
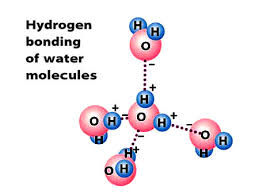
Hydrogen Bonding- the week attraction between the weakly positive hydrogen atoms and the weakly negative oxygen atoms of a different molecule. Water is a solvent that attracted other molecules that can create a film on top of the surfaces of bodies of water.
States of Matter- Earth is the only planet in the solar system where water is found in liquid water and one calorie is equal to the amount of heat needed to raise the temperature of 1 gram of water by 1.